Rate of Reaction
The rate of reaction is the change in concentration of a reactant or product per unit of time. The rate of a reaction can not be measured directly, however we can use the following formula to calculate the change in reactant or product in a certain amount of time:
The rate of reaction is the change in concentration of a reactant or product per unit of time. The rate of a reaction can not be measured directly, however we can use the following formula to calculate the change in reactant or product in a certain amount of time:
There are several factors which affect the rate of a chemical reaction. These factors are:
If the temperature is increased the speed of the molecules is increased. This in turn increases the energy of the molecules and the number of successful collisions; which increases the rate.
Concentration
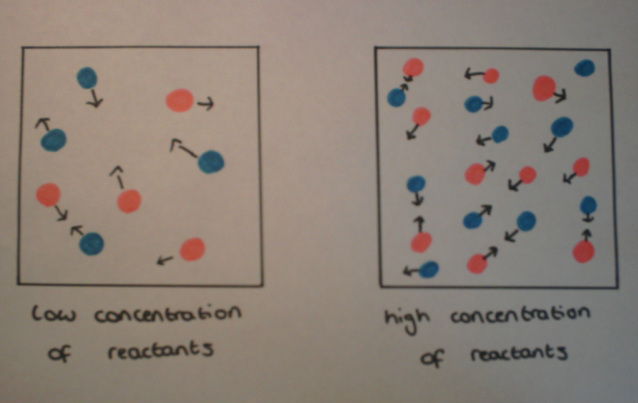
When the concentration of a solution is increased, this increases the number of reacting molecules in the given volume. This means that collisions are more likely to occur which increases the chance of successful collisions and increases the rate of reaction.
The diagram below shows how there is a greater chance of collisions when the concentration of a solution is increased.
- temperature
- concentration
- the presence of a catalyst
- surface area (solids)
- pressure (gases)
If the temperature is increased the speed of the molecules is increased. This in turn increases the energy of the molecules and the number of successful collisions; which increases the rate.
Concentration
When the concentration of a solution is increased, this increases the number of reacting molecules in the given volume. This means that collisions are more likely to occur which increases the chance of successful collisions and increases the rate of reaction.
The diagram below shows how there is a greater chance of collisions when the concentration of a solution is increased.
The presence of a catalyst
A catalyst is a substance which increases the rate of a chemical reaction with out being used up. It achieves this by finding an alternative pathway with a lower activation energy for the reaction.
Surface area
If the surface area of a solid reactant is increased this means that there are more molecules available to react. As a result of this, due to a higher chance of collisions, the rate of reaction is increased.
Pressure
By increasing the pressure of a gas, there is more molecules in the given volume, so this causes more collisions to occur thus increasing the rate of reaction.
In my investigation I will be looking at how temperature and concentration affects the rate of the two-colour chemiluminescent clock reaction.
A catalyst is a substance which increases the rate of a chemical reaction with out being used up. It achieves this by finding an alternative pathway with a lower activation energy for the reaction.
Surface area
If the surface area of a solid reactant is increased this means that there are more molecules available to react. As a result of this, due to a higher chance of collisions, the rate of reaction is increased.
Pressure
By increasing the pressure of a gas, there is more molecules in the given volume, so this causes more collisions to occur thus increasing the rate of reaction.
In my investigation I will be looking at how temperature and concentration affects the rate of the two-colour chemiluminescent clock reaction.