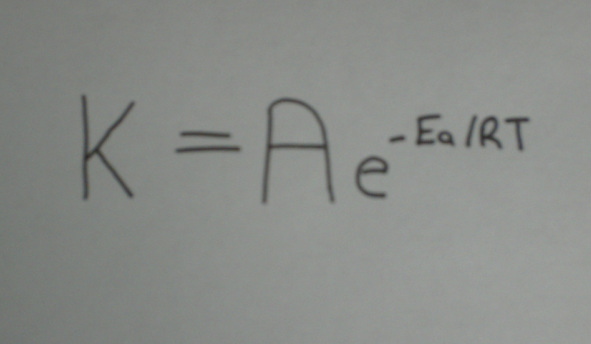The Arrhenius Equation
The Arrhenius equation is a mathematical expression which links the rate of constant of a chemical reaction and the exponential value of temperature.
The Arrhenius equation is a mathematical expression which links the rate of constant of a chemical reaction and the exponential value of temperature.
Where:
- T is temperature in kelvin (K)
- R is the gas constant. The gas constant comes from the equation pV=nRT which relates the pressure, volume and temperature of a particular number of moles of a gas.
- Ea is the activation energy
- e is a mathematical number with the value of 2.71828
- A is the frequency factor (also known as the pre-exponential factor. It takes into account factors such as the frequency of collisions and often varies with temperature.
I can use the Arrhenius equation to calculate and show how the rate constant changes after the temperature has been changed.