Background Information
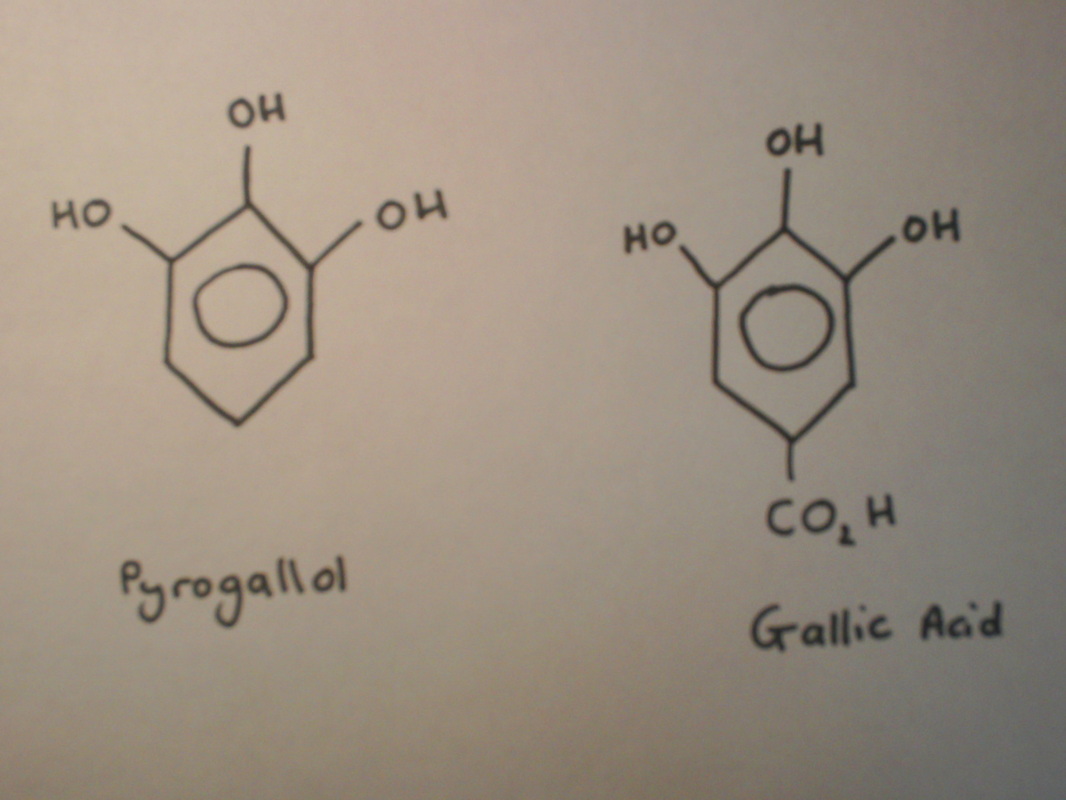
In the two-colour chemiluminescent clock reaction the pyrogallol and formaldehyde reacts with hydrogen peroxide to generate an oxygen molecule with the electrons paired together, this is know as a singlet oxygen. The singlet oxygen is then converted into an oxygen molecule with the electrons unpaired; this is known as a triplet oxygen. This releases a weak emission of red light. Heat is also produced during this reaction and the oxygen gas causes the bubbling.
Research carried out about this mechanism has used gallic acid to replace pyrogallol because it provides more reliable results. However the understanding of the reaction can be directly applied to the reaction using pyrogallol.
In the two-colour chemiluminescent clock reaction the pyrogallol and formaldehyde reacts with hydrogen peroxide to generate an oxygen molecule with the electrons paired together, this is know as a singlet oxygen. The singlet oxygen is then converted into an oxygen molecule with the electrons unpaired; this is known as a triplet oxygen. This releases a weak emission of red light. Heat is also produced during this reaction and the oxygen gas causes the bubbling.
Research carried out about this mechanism has used gallic acid to replace pyrogallol because it provides more reliable results. However the understanding of the reaction can be directly applied to the reaction using pyrogallol.
Formaldehyde reacts with the hydroperoxide ion to produce compounds A and B, (please see slideshow below). Further oxidation can lead to carbon dioxide, water and formic acid being produced.
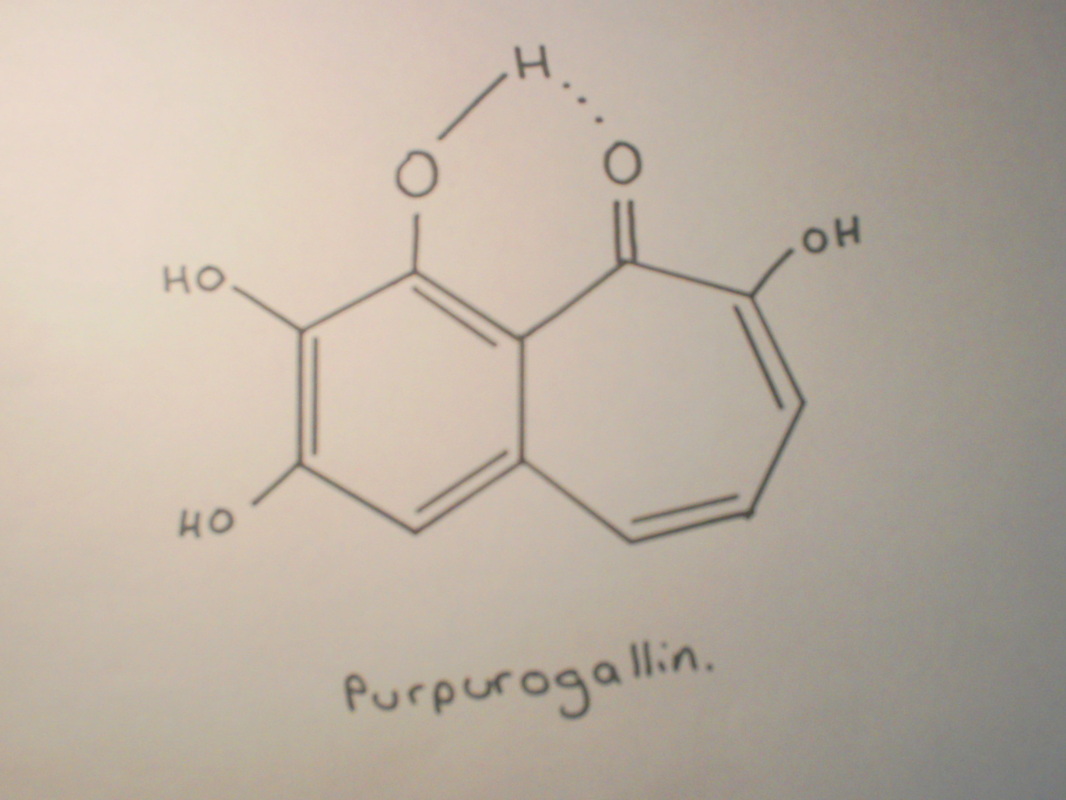
The oxidation of pyrogallol is more complex than that of formaldehyde because it does not result in the same product each time. The most common product is purpurogallin.
The mechanism which has been proposed for the production of purpurogallin by the oxidation of pyrogallol is shown below.
The luminol reacts by loosing a N2 molecule, forming a product in an excited state which emits blue light. This reaction is triggered by the red emission of light; which suggests that there are free radicals involved in this reaction.